
 | Laboratories \ Laboratory for Ecological Monitoring of Nuclear Power Plant (NPP) Regions and bioindication \ Scientific Staff \ Biazrov Lev G. |
| Goals and Tasks |  | Publications |  | Scientific Staff |
 |
Changes of Epiphytic Lichen Species Diversity of Moscow City –
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| VII | VIII | XV | XVI | XXIII | XXIV | ||||||
| 1990 | 2006 | 1990 | 2006 | 1990 | 2006 | 1990 | 2006 | 1990 | 2006 | 1990 | 2006 |
| 6 | 14 | ||||||||||
| 9 | 16 | 9 | 15 | ||||||||
| 2 | 6 | 0 | 10 | ||||||||
| 6 | 7 | 7 | 14 | 1 | 4 | 0 | 4 | 7 | 10 | ||
| 3 | 7 | 5 | 17 | 1 | 6 | 0 | 5 | 13 | 17 | 10 | 20 |
| 0 | 5 | 0 | 8 | 0 | 8 | 0 | 10 | 10 | 17 | 11 | 17 |
| 3 | 7 | 5 | 7 | 0 | 6 | 0 | 8 | 10 | 20 | 10 | 20 |
| 3 | 8 | 2 | 10 | 0 | 8 | 4 | 18 | 11 | 20 | 12 | 18 |
| 4 | 12 | 2 | 10 | 4 | 15 | 6 | 12 | 12 | 22 | 8 | 19 |
| 7 | 15 | 7 | 11 | 4 | 7 | 5 | 12 | 6 | 16 | 11 | 17 |
| 7 | 12 | 7 | 12 | 0 | 3 | 2 | 11 | 5 | 17 | 8 | 11 |
| 5 | 18 | 8 | 19 | 2 | 9 | 0 | 5 | 2 | 14 | 0 | 5 |
| 1 | 14 | 2 | 14 | 0 | 2 | 0 | 6 | 1 | 14 | 2 | 15 |
| 0 | 9 | 1 | 11 | 1 | 7 | 0 | 9 | 0 | 11 | 0 | 8 |
| 0 | 12 | 4 | 12 | 0 | 2 | 1 | 10 | 0 | 9 | 0 | 8 |
| 1 | 15 | 0 | 13 | 1 | 6 | 0 | 7 | 0 | 7 | 0 | 12 |
| 4 | 13 | 5 | 16 | 0 | 8 | 1 | 10 | 0 | 9 | 0 | 10 |
| 5 | 12 | 9 | 16 | 1 | 9 | 0 | 5 | 0 | 7 | 0 | 6 |
| 18 | 26 | 11 | 17 | 1 | 3 | 1 | 7 | 0 | 5 | 0 | 4 |
| 3 | 13 | 7 | 13 | 0 | 3 | 0 | 4 | 0 | 4 | 0 | 9 |
| 2 | 10 | 14 | 21 | 1 | 7 | 0 | 8 | 0 | 11 | 0 | 4 |
| 4 | 11 | 9 | 19 | 1 | 9 | 0 | 7 | 0 | 8 | 0 | 8 |
| 2 | 12 | 2 | 10 | 1 | 10 | 0 | 5 | 0 | 9 | 0 | 11 |
| 4 | 10 | 3 | 11 | 0 | 5 | 0 | 9 | 1 | 10 | 0 | 8 |
| 0 | 8 | 0 | 14 | 0 | 8 | 0 | 10 | 0 | 9 | 0 | 11 |
| 4 | 14 | 3 | 15 | 4 | 10 | 3 | 8 | 0 | 10 | 0 | 14 |
| 2 | 15 | 4 | 13 | 3 | 10 | 3 | 9 | 0 | 9 | 0 | 9 |
| 0 | 9 | 4 | 15 | 0 | 12 | 4 | 12 | 0 | 7 | 0 | 8 |
| 0 | 14 | 4 | 16 | 0 | 12 | 0 | 13 | 0 | 6 | 0 | 5 |
| 2 | 13 | 2 | 14 | 6 | 13 | 3 | 16 | 0 | 8 | 0 | 6 |
| 9 | 20 | 7 | 25 | 3 | 12 | 2 | 11 | 0 | 9 | 0 | 7 |
| 3 | 20 | 10 | 16 | 2 | 9 | 5 | 15 | 0 | 9 | 0 | 9 |
| 0 | 13 | 12 | 21 | 3 | 10 | 4 | 10 | 0 | 8 | 0 | 7 |
| 1 | 13 | 5 | 9 | 3 | 9 | 0 | 14 | 0 | 6 | ||
| 0 | 7 | 2 | 14 | 2 | 9 | 3 | 9 | 7 | 17 | ||
| 2 | 9 | 0 | 9 | 5 | 14 | 5 | 24 | ||||
| 5 | 15 | 0 | 6 | ||||||||
| 5 | 11 | 0 | 7 | ||||||||
Western transect (columns VII and VIII in width 1 km everyone in tab. 1) in North began at crossing the October railway with MRMW, crossed parts of forest parks Pokrovskoe-Streshnevo and Fili-Kuntsevo, and in the South came to an end on MRMW in area Teplyi Stan. Central transect (columns XV and XVI) in North began from crossing Altuf'evo highway with MRMW, crossed a part of territory of the Main Botanical Garden, of the Alexandrovskij garden in city centre and in the South came to an end at crossing the Warsaw highway with MRMW. East transect (columns XXIII and XXIV) in North began from crossing the Yaroslavl highway with MRMW, passed through the forest park Losinyi Ostrov, the area Mar'ino and in the South came to an end to the east of crossing the Kashira highway with MRMW.
| 10 | 2006 | 10 | 8 | 6 | 10 | 15 | 11 | 7 | 8 | 9 | 18 | 10 | 3 | 7 | 12 | 16 | 15 | 9 | 18 | 20 | 17 | 16 | 17 | 22 | 18 | 20 | 15 | 18 | |||
| 1990 | 1 | 1 | 1 | 2 | 7 | 7 | 0 | 1 | 2 | 7 | 2 | 0 | 4 | 5 | 4 | 2 | 0 | 4 | 5 | 3 | 6 | 11 | 12 | 10 | 10 | 11 | 9 | ||||
| 11 | 2006 | 8 | 18 | 10 | 2 | 11 | 12 | 12 | 8 | 6 | 6 | 14 | 12 | 4 | 3 | 11 | 10 | 12 | 15 | 21 | 22 | 22 | 17 | 11 | 12 | 15 | 26 | 19 | 11 | 12 | |
| 1990 | 5 | 5 | 2 | 0 | 3 | 7 | 7 | 2 | 0 | 4 | 10 | 10 | 3 | 0 | 2 | 3 | 3 | 0 | 7 | 6 | 2 | 5 | 8 | 3 | 7 | 14 | 4 | 0 | 5 | ||
| 19 | 2006 | 17 | 20 | 15 | 19 | 12 | 12 | 26 | 17 | 11 | 7 | 9 | 11 | 8 | 10 | 3 | 7 | 3 | 9 | 8 | 9 | 2 | 6 | 5 | 4 | 8 | 10 | 15 | 14 | 15 | 12 |
| 1990 | 7 | 10 | 7 | 8 | 4 | 4 | 18 | 11 | 3 | 3 | 2 | 2 | 2 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 2 | 3 | |
| 20 | 2006 | 19 | 17 | 12 | 14 | 8 | 18 | 13 | 13 | 10 | 14 | 5 | 6 | 7 | 11 | 3 | 4 | 5 | 4 | 9 | 6 | 7 | 7 | 4 | 9 | 10 | 13 | 14 | 17 | 11 | 8 |
| 1990 | 13 | 10 | 0 | 4 | 4 | 9 | 3 | 7 | 6 | 3 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 4 | 4 | 1 | 2 | |
| 28 | 2006 | 9 | 22 | 15 | 9 | 15 | 11 | 14 | 17 | 10 | 14 | 14 | 12 | 12 | 10 | 11 | 12 | 9 | 4 | 7 | 7 | 8 | 5 | 3 | 2 | 14 | 17 | 17 | |||
| 1990 | 2 | 17 | 4 | 0 | 4 | 4 | 10 | 10 | 3 | 3 | 4 | 0 | 4 | 0 | 0 | 8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 6 | 5 | 3 | ||||
| 29 | 2006 | 22 | 18 | 13 | 16 | 23 | 12 | 10 | 11 | 13 | 17 | 12 | 13 | 9 | 12 | 16 | 3 | 10 | 8 | 6 | 5 | 6 | 4 | 4 | 14 | 22 | 19 | ||||
| 1990 | 16 | 12 | 0 | 4 | 10 | 2 | 2 | 4 | 6 | 2 | 0 | 0 | 0 | 0 | 5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 5 | 9 | 10 |
Northern transect (lines 10 and 11 in tab. 2) in West began from MRMW in area Strogino (Isakovsky street), crossed the Forest trial plot of Timiryazev agricultural academy, a southern part of Losinyi Ostrov forest park and in East came to an end at MRMW in area Gol'yanovo (Krasnoyarsk street). Central transect (lines 19 and 20) in West began from MRMW to the north of Molodogvardeiskaya street, and in East came to an end at a place of crossing MRMW with railway in Vladimir city. Southern transect (lines 28 and 29) in West began from crossing the Borovsk highway with MRMW, and in East crossed a southern part of Kuz'minki forest park and came to an end at MRMW to the north of the Verkhnie Polya street.
Total number of the surveyed squares 1 x 1 km has made 336, i.e. more thirds of all area of city in borders of MRMW.
In all in 2006 in these 336 squares have been found out representatives of 64 epiphytic lichen species living on trunks of trees and bushes up to height of 2.5 m (tab. 3). In table 3 in addition to lichen species composition recorded on surveyed transects in 1988-1991 and 2006, are shown: presence of bodies of propagulation – A – apothecia, I – isidia, S – soredia; the relation of a species to value pH of substratum – R (R1 – a substratum extremely acid and very acid, pH <4.0; R2 – a substratum rather acid, pH 4.1-4.8; R3 – a substratum moderately acidr, pH 4.9-5.6; R4 – a substratum semi-neutral, pH 5.7-6.5; R5 – a substratum neutral or alkaline, pH> 6.5); the relation of a species to riches of substratum by elements of nutrients – N (N1 – very poor by mineral elements (Mg, Ca, K, Na, N) bark of trees as at a spruce, larch, birch; N2 – moderately rich by mineral elements bark of trees with very small eutrophication; N3 – the bark is rich with mineral elements or it is moderately covered by a dust; N4 – the bark rich with mineral elements, it is often covered by a dense layer of a dust); the relation of a species to a degree of air pollution of territory – toxiphoby (To) – To1 – the most tolerant to air pollution species, To5 – the species, most sensitive to air pollution, To? – data on reaction of a species to air pollution for the author are unknown. The relation of a species to the factors of environment are assessed by five-ball scales on the basis of updating already of available generalizations (Insarova, Insarov, 1989; Wirth, 1991) and first hand (Biazrov, 1994, 2002). The representative of group To5 in Moscow are not found out yet.
| Propagulation bodies 1 | Species | 1990 | 2006 | Ecological characteristic of species 1 | ||||
| Q | % | Q | % | R | N | To | ||
| A | Biatora helvola Körb. ex Hellb. | 0 | 0 | 54 | 16.1 | R3 | N1 | To4 |
| A | Buellia punctata (Hoffm.) A. Massal.= Amandinea punctata (Hoffm.) Coppins & Scheid. | 0 | 0 | 2 | 0.6 | R3 | N3 | To2 |
| A | Caloplaca cerina (Ehrh. ex Hedwig) Th. Fr. | 8 | 2.4 | 189 | 56.2 | R4 | N3 | To2 |
| A | C. holocarpa (Hoffm. ex Ach.) Wade | 11 | 3.3 | 107 | 31.8 | R5 | N3 | To3 |
| A | Candelariella vitellina (Hoffm.) Müll. Arg. | 18 | 5.4 | 192 | 57.1 | R3 | N3 | To2 |
| A | C. xanthostigma (Ach.) Lettau | 0 | 0 | 3 | 0.9 | R3 | N3 | To3 |
| A | Cetraria sepincola (Ehrh.) Ach. | 10 | 3 | 17 | 5.1 | R2 | N2 | To4 |
| S | Chrysothrix candelaris (L.) J. R. Laundon | 0 | 0 | 18 | 5.3 | R2 | N2 | To4 |
| A | Cladonia caespitica (Pers.) Flörke | 1 | 0.3 | 0 | 0 | R3 | N2 | To4 |
| A,S | C. cenotea (Ach.) Schaer | 0 | 0 | 1 | 0.3 | R1 | N1 | To3 |
| A,S | C. chlorophaea (Flörke ex Sommerf.) Spreng. | 16 | 4.7 | 2 | 0.6 | R2 | N2 | To4 |
| A,S | C. coniocraea (Flörke) Spreng. | 54 | 16.1 | 71 | 21.1 | R2 | N2 | To3 |
| A,S | C.digitata (L.) Hoffm. | 5 | 1.5 | 0 | 0 | R1 | N2 | To2 |
| A,S | C. fimbriata (L.) Fr. | 38 | 11.3 | 66 | 19.6 | R2 | N1 | To3 |
| A,S | C. glauca Flörke | 0 | 0 | 1 | 0.3 | R1 | N1 | To? |
| A,S | C. macilenta Hoffm. | 0 | 0 | 1 | 0.3 | R1 | N1 | To? |
| A,S | C. ochrochlora Flörke | 0 | 0 | 1 | 0.3 | R1 | N2 | To4 |
| A,S | C. ramulosa (With.) J. R. Laundon | 0 | 0 | 1 | 0.3 | R1 | N2 | To? |
| A | C. squamosa Hoffm. | 0 | 0 | 1 | 0.3 | R1 | N2 | To? |
| A,S | C. subulata (L.) Weber ex F. H. Wigg. | 0 | 0 | 4 | 1.2 | R2 | N1 | To? |
| S | Evernia prunastri (L.) Ach. | 0 | 0 | 1 | 0.3 | R2 | N3 | To4 |
| S | Hypocenomyce scalaris (Ach.) M. Choisy | 14 | 4.2 | 32 | 9.5 | R1 | N2 | To3 |
| S | Hypogymnia physodes (L.) Nyl. | 80 | 23.8 | 120 | 35.7 | R2 | N2 | To3 |
| A | Lecania dubitans (Nyl.) A. L. Sm. | 0 | 0 | 2 | 0.6 | R2 | N2 | To3 |
| A | L. fuscella (Schaer.) A. Massal. | 0 | 0 | 5 | 1.5 | R3 | N2 | To3 |
| A | Lecanora carpinea (L.) Vain. | 0 | 0 | 2 | 0.6 | R3 | N2 | To4 |
| A | L. hagenii (Ach.) Ach. | 27 | 8 | 221 | 65.8 | R5 | N4 | To1 |
| A | L. piniperda Körb. | 0 | 0 | 32 | 9.5 | R1 | N2 | To3 |
| A | L. pulicaria (Pers.) Ach. | 0 | 0 | 4 | 1.2 | R1 | N2 | To3 |
| A | L. symmicta (Ach.) Ach. | 1 | 0.3 | 12 | 3.6 | R3 | N3 | To4 |
| A | L. varia (Hoffm.) Ach. | 27 | 8 | 84 | 25 | R2 | N2 | To2 |
| A | Lecidea erythrophaea Flörke ex Sommerf. | 2 | 0.6 | 9 | 2.7 | R3 | N1 | To4 |
| A | Lecidella elaeochroma (Ach.) M. Choisy | 0 | 0 | 1 | 0.3 | R3 | N3 | To3 |
| S | Lepraria incana (L.) Ach. | 33 | 9.8 | 71 | 21.1 | R2 | N2 | To1 |
| S | L. lobificans Nyl. | 0 | 0 | 7 | 2 | R2 | N2 | To4 |
| A | Maronea constans (Nyl.) Hepp | 0 | 0 | 1 | 0.3 | R2 | N2 | To? |
| I | Melanelia elegantula (Zahlbr.) Essl. | 0 | 0 | 1 | 0.3 | R2 | N2 | To4 |
| I | M. exasperata (De Not.) Essl | 1 | 0.3 | 5 | 1.5 | R3 | N3 | To4 |
| I | M.exasperatula (Nyl.) Essl. | 1 | 0.3 | 8 | 2.4 | R3 | N3 | To3 |
| A | M. olivacea (L.) Essl. | 0 | 0 | 18 | 5.3 | R1 | N2 | To3 |
| I,S | M.subargentifera (Nyl.) Essl. | 0 | 0 | 1 | 0.3 | R4 | N4 | To4 |
| A | Micarea lignaria (Ach.) Hedl. | 0 | 0 | 4 | 1.2 | R2 | N2 | To2 |
| A | Opegrapha atra Pers. | 39 | 11.6 | 21 | 6.3 | R3 | N3 | To4 |
| A | O. rufescens Pers. | 2 | 0.6 | 111 | 33 | R3 | N3 | To4 |
| S | Parmelia sulcata Taylor | 111 | 33 | 218 | 64.9 | R3 | N3 | To2 |
| S | Parmeliopsis ambigua (Wulfen) Nyl. | 3 | 0.9 | 0 | 0 | R1 | N2 | To4 |
| S | P. hyperopta (Ach.) Arnold | 1 | 0.3 | 0 | 0 | R1 | N2 | To3 |
| S | Pertusaria albescens (Hudson) M. Choisy & Werner | 0 | 0 | 2 | 0.6 | R3 | N3 | To4 |
| A | P. leioplaca DC. | 0 | 0 | 2 | 0.6 | R3 | N2 | To3 |
| A | Phaeophyscia ciliata (Hoffm.) Moberg | 3 | 0.9 | 0 | 0 | R3 | N3 | To2 |
| S | P. nigricans (Flörke) Moberg | 4 | 1.2 | 24 | 7.1 | R5 | N4 | To2 |
| S | P. orbicularis (Neck.) Moberg | 145 | 43.1 | 336 | 100 | R4 | N4 | To1 |
| S | P. sciastra (Ach.) Moberg | 0 | 0 | 179 | 53.3 | R5 | N4 | To2 |
| S | Physcia adscendens (Fr.) H. Oliver | 0 | 0 | 272 | 81 | R4 | N4 | To2 |
| A | P. aipolia (Ehrh.ex Humb.) Fürnr. | 1 | 0.3 | 0 | 0 | R4 | N3 | To4 |
| A | P. stellaris (L.) Nyl. | 161 | 47.9 | 307 | 91.4 | R3 | N3 | To1 |
| S | P. tenella (Scop.) DC. | 0 | 0 | 8 | 2.4 | R3 | N4 | To2 |
| S | P. tribacia (Ach.) Nyl. | 5 | 1.5 | 94 | 28.0 | R3 | N3 | To2 |
| A | Physconia distorta (With.) J. R. Laundon | 0 | 0 | 1 | 0.3 | R4 | N4 | To3 |
| I,S | P. grisea (Lam.) Poelt | 1 | 0.3 | 0 | 0 | R4 | N4 | To2 |
| S | Ramalina farinacea (L.) Ach. | 0 | 0 | 1 | 0.3 | R3 | N2 | To3 |
| S | R. pollinaria (Westr.) Ach. | 0 | 0 | 1 | 0.3 | R2 | N3 | To4 |
| A | Rinodina pyrina (Ach.) Arnold | 0 | 0 | 6 | 1.8 | R4 | N3 | To3 |
| A,S | Scoliciosporum chlorococcum (Graewe ex Stenh.) Vezda | 138 | 41 | 336 | 100 | R2 | N3 | To1 |
| A | Strangospora pinicola (A. Massal.) Körb.2 | 0 | 0 | 4 | 1.2 | R2 | N3 | To1 |
| S | Trapeliopsis granulosa (Hoffm.) Lumbsch | 0 | 0 | 34 | 10.1 | R2 | N2 | To1 |
| S | Tuckermannopsis chlorophylla (Willd.) Hale | 1 | 0.3 | 2 | 0.6 | R2 | N2 | To4 |
| S | Vulpicida pinastri (Scop.) J.-E. Mattsson & M. J. Lai | 30 | 8.9 | 41 | 12.2 | R1 | N1 | To3 |
| S | Xanthoria candelaria (L.) Th. Fr. | 0 | 0 | 2 | 0.6 | R3 | N4 | To3 |
| A | X. parietina (L.) Th. Fr. | 35 | 10.4 | 326 | 97.0 | R4 | N4 | To1 |
| A | X. polycarpa (Hoffm.) Th. Fr. ex Rieber | 2 | 0.6 | 57 | 17.0 | R2 | N4 | To2 |
| Total species | 35 | 64 | ||||||
1 – explanation of symbols is given in the text
2 – this species before in territory of city was found by E.E. Muchnik that is marked in the WEB-publication:
Biazrov L.G. 2006. Checklist of the lichen biota of the Moscow region (Russia) //
http://www.sevin.ru/laboratories_eng/biazrov_msk.html
In 2006 lichens have been recorded in all 336 surveyed squares 1 x 1 km, the number of species per a square varied from 2 up to 26 at average value of this parameter about 11 (tab. 4). The species Evernia prunastri, Ramalina farinacea, R. pollinaria considered as sensitive to acid pollution (Wirth, 1991), are recorded only one time and have been submitted small (up to 2 cm) by the sickly specimens growing on the bases of trunks of trees in forest tracts.
| Number of species per a square | 1990 | 2006 | ||
| Number of squares | % | Number of squares | % | |
| 0 | 125 | 37.2 | 0 | 0 |
| 1 | 30 | 8.9 | 0 | 0 |
| 2 | 36 | 10.7 | 5 | 1.5 |
| 3 | 28 | 8.3 | 8 | 2.4 |
| 4 | 29 | 8.6 | 11 | 3.3 |
| 5 | 20 | 5.9 | 12 | 3.6 |
| 6 | 9 | 2.7 | 17 | 5.1 |
| 7 | 14 | 4.2 | 23 | 6.8 |
| 8 | 5 | 1.5 | 28 | 8.3 |
| 9 | 8 | 2.4 | 35 | 10.4 |
| 10 | 15 | 4.5 | 31 | 9.2 |
| 11 | 5 | 1.5 | 21 | 6.3 |
| 12 | 5 | 1.5 | 25 | 7.4 |
| 13 | 2 | 0.6 | 12 | 3.6 |
| 14 | 2 | 0.6 | 24 | 7.1 |
| 15 | 0 | 0 | 18 | 5.4 |
| 16 | 1 | 0.3 | 9 | 2.7 |
| 17 | 1 | 0.3 | 16 | 4.7 |
| 18 | 1 | 0.3 | 10 | 3 |
| 19 | 0 | 7 | 2 | |
| 20 | 0 | 9 | 2.7 | |
| 21 | 0 | 3 | 0.9 | |
| 22 | 0 | 7 | 2 | |
| 23 | 0 | 1 | 0.3 | |
| 24 | 0 | 1 | 0.3 | |
| 25 | 0 | 1 | 0.3 | |
| 26 | 0 | 2 | 0.6 | |
| Average number of species per a square | 3.1 | 11.3 | ||
16 years ago on these six transects in the same level of settlement on trunks of trees have been recorded representatives of 35 lichen species (tab. 3). They then were absent in 125 squares 1 x 1 km (37 % from the total number). From 64 species which recorded in 2006 only representatives of two – Phaeophyscia orbicularis and Scoliciosporum chlorococcum – are marked in all 336 squares 1 x 1 km. 8 species have recorded been in more than 50 % of total number of squares – Caloplaca cerina (56 %), Candelariella vitellina (57 %), Lecanora hagenii (66 %), Parmelia sulcata (65 %), Phaeophyscia sciastra (53 %), Physcia adscendens (81 %), Physcia stellaris (91 %), Xanthoria parietina (97 %). At 11 species in 2006 index frequency in squares of all transects was from 10 up to 50 %. Almost third of species in 2006 have been recorded only in 1-2 squares.
According to registers of 1988-1991 were absent species, which the index frequency on the same six transects exceeded 50 %, (tab. 3). Those years the representatives only three lichen species have been found out more than in 40% of these surveyed squares – Phaeophyscia orbicularis (43 %), Physcia stellaris (49 %), Scoliciosporun chlorococcum (41 %). To 2006 the same species, and also Xanthoria parietina, Physcia adscendens have occupied all or nearly so all squares of these of six transects under study.
Among the species recorded in the end 1980th and in 2006 quantitatively predominate the species preferring rather acid (pH 4.1-4.8) and moderately acid (pH 4.9-5.6) substratum (tab. 5).
| The rank of pH value (R)1 | 1990 | 2006 | ||
| Number of species | % | Number of species | % | |
| R1 | 5 | 14.3 | 11 | 17.2 |
| R2 | 10 | 28.6 | 21 | 32.8 |
| R3 | 12 | 34.2 | 21 | 32.8 |
| R4 | 5 | 14.3 | 7 | 10.9 |
| R5 | 3 | 8.6 | 4 | 6.3 |
1 – explanation of symbols is given in the text
However the species having high value of frequency [Caloplaca cerina (R4), C. holocarpa (R5), Lecanora hagenii (R5), Parmelia sulcata (R3), Phaeophyscia orbicularis (R4), P. sciastra (R5), Physcia adscendens (R4), P. stellaris (R3), Xanthoria parietina (R4)], more often prefer semi-neutral or neutral substrata (tab. 6). As a whole in both of terms more than half of species concerned to a rank of frequency <5 % (tab. 6).
| The rank of pH value (R)1 | Year | The rank of frequency of species (% of squares) | Number of species | ||||
| <5% | 5.1-15 | 15.1-30 | 30.1-50 | >50% | |||
| R1 | 1990 | 4 | 1 | 0 | 0 | 0 | 5 |
| 2006 | 6 | 4 | 1 | 0 | 0 | 11 | |
| R2 | 1990 | 4 | 4 | 1 | 1 | 0 | 10 |
| 2006 | 11 | 3 | 5 | 1 | 1 | 21 | |
| R3 | 1990 | 8 | 2 | 0 | 2 | 0 | 12 |
| 2006 | 13 | 2 | 2 | 1 | 3 | 21 | |
| R4 | 1990 | 3 | 1 | 0 | 1 | 0 | 5 |
| 2006 | 3 | 0 | 0 | 0 | 4 | 7 | |
| R5 | 1990 | 2 | 1 | 0 | 0 | 0 | 3 |
| 2006 | 0 | 1 | 0 | 0 | 3 | 4 | |
| Total | 1990 | 21 | 9 | 1 | 4 | 0 | 35 |
| 2006 | 33 | 10 | 8 | 2 | 11 | 64 | |
1 – explanation of symbols is given in the text
Among epiphytic lichens of city on number of species predominate such which settle on moderately rich mineral elements bark of trees with very small eutrophication or on bark which rich mineral elements or moderately covered by a dust (tab. 7). However species with high index frequency as Lecanora hagenii (N4), Phaeophyscia orbicularis (N4), P. sciastra (N4), Physcia adscendens (N4), Xanthoria parietina (N4) they characterize as preferring substrata which are rich by elements of nutrients (Wirth, 1991).
| The rank of riches substratum by nutrients (N)1 | 1990 | 2006 | ||
| Number of species | % | Number of species | % | |
| N1 | 3 | 8.6 | 8 | 12.5 |
| N2 | 12 | 34.3 | 25 | 39.1 |
| N3 | 14 | 40 | 20 | 31.2 |
| N4 | 6 | 17.1 | 11 | 17.2 |
1 – explanation of symbols is given in the text
The comparison of species on a degree of stability to pollution shows that for the period from 1988-1991 to 2006 there was an remarkable increase of number of species rather more sensitive to air pollution (classes To3 and To4) though the quota of species of a class To4 in total number recorded taxa has remained almost at a former level (tab. 8).
| The rank of toxiphoby (To)1 | 1990 | 2006 | ||
| Number of species | % | Number of species | % | |
| To1 | 4 | 11.4 | 6 | 9.4 |
| To2 | 10 | 28.6 | 13 | 20.3 |
| To3 | 8 | 22.9 | 19 | 29.7 |
| To4 | 11 | 31.4 | 19 | 29.7 |
| To? | 2 | 5.7 | 7 | 10.9 |
1 – explanation of symbols is given in the text
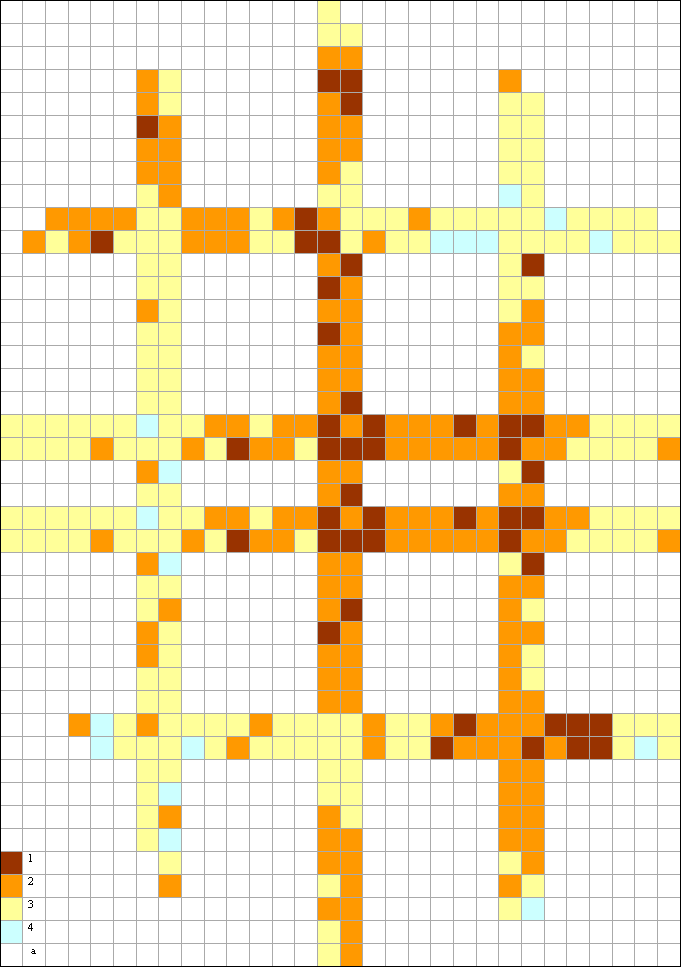 |
Fig. 1 The scheme of division of sites on transects through territory of Moscow in 2006 on number of epiphytic lichen species per 1 km2: 1 – critically dangerous sites (2-5 species); 2 – extremely dangerous sites (6-10 species); 3 – dangerous sites (11-20 species); 4 – moderately dangerous sites (>20 species); a – side of a square is 1 km |
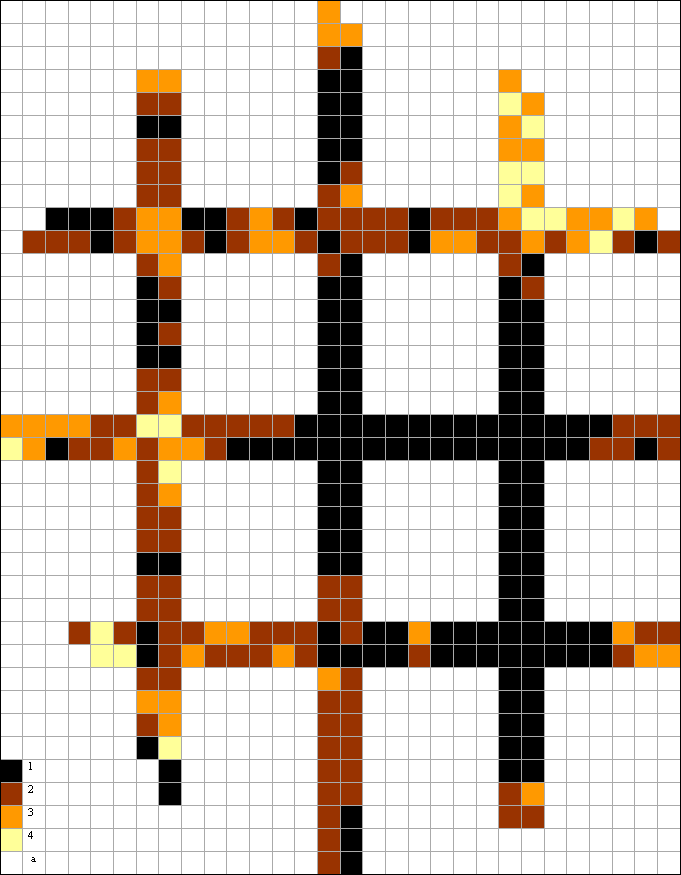 |
Fig. 2 The scheme of division of sites on transects through territory of Moscow per 1988-1991 on number of epiphytic lichen species per 1 km2: 1 – "lichen desert" (0-1 species), catastrophically dangerous sites; 2 – critically dangerous sites (2-5 species); 3 – extremely dangerous sites (6-10 species); 4 – dangerous sites (>10 species); a – side of a square is 1 km |
Formerly it has been shown that by results of the investigation of features of epiphytic lichens distribution per 1988-1991 on number of species per a square was carried out ranking of city territory on a level of air pollution. The similar way (on number of species per a square) has been applied for graphic display of results received in 2006 (fig. 1). The grouping of squares on tansects was similar to what has been used in the beginning 1990th. Also former was the approach to having filled of squares – from dark color to light that corresponded to transition from unfavorable conditions of life for lichens to more favorable.
The comparison with a clipping of the same transects (fig. 2) from corresponding skeleton map of a beginning 1990th (Biazrov, 1996) shows that in the surveyed territory in 2006 the situation for lichens has cardinally improved.
The rank of skeleton map legend "lichen desert" has disappeared but has appeared rank "more than 20 species". On third has decreased the quota of rank "2-5 species", more than in 2 times has increased the quota of rank "6-10 species " and more than in 8 times the quota of rank "11-20 species" (tab. 9).
By 2006 the number of epiphytic lichen species has increased in all surveyed squares 1 x 1 km, as in the central part of city and its peripheral parts. It allows to make a conclusion that in comparison with the beginning 1990 the conditions for ability to live of a number of lichen species were improved. It is expressed not only in increase of number of species per unit of the area, but also in increase of index frequency on the areas under study of the representatives Caloplaca cerina, Candelariella vitellina, Phaeophyscia orbicularis, Physcia stellaris, Scoliciosporum chlorococcum, Xanthoria parietina and some other species, at that the thalli Phaeophyscia orbicularis covered significant surfaces of trunks of trees quite often.
| The contour of legends on number of species per a square | 1990 | 2006 | ||
| Number of squares | % | Number of squares | % | |
| 0-1 | 155 | 46.1 | 0 | 0 |
| 2-5 | 113 | 33.5 | 36 | 10.7 |
| 6-10 | 51 | 15.3 | 134 | 39.9 |
| 11-20 | 17 | 5.1 | 151 | 44.9 |
| >20 | 0 | 0 | 15 | 4.5 |
| Total | 336 | 100 | 336 | 100 |
Dominating over ecology anthropocentrism when the man is separated from nature, and its activity is considered as external concerning natural processes, puts in the face of the researcher at the analysis of data about parameters of the biological objects received in one place, but in different years, an additional problem which consists in necessity to isolate from a total sum of the revealed changes what are connected with natural processes, and what are caused by activity of the man to which they concerns the pollution of environment also. It is known that in natural communities takes place the continuous process of their development which is caused as interrelation of organisms with each other and with environment changed by them that usually name succession, and influence of the external factors in relation to community including activity of the man named as exogenous changes (Rabotnov, 1983). The established changes of lichen species composition and communities formed by them which have occurred for a known interval of time, undoubtedly, are a consequence that and another. Also it is necessary clearly to realize that the control over dynamics of lichen species composition at monitoring of environment provide faster correlations than proofs of causal connections, except for cases when these investigations are a part of controllable reproduced experiments, at which reconstruct those or other parameters of air quality. But such experiences are not enough, and we are compelled to do conclusions being based on the published data on reaction of different lichen species on pollutants of air and own experience. These conclusions should help to isolate from a total sum of the revealed changes what are connected with effect of pollutants. The arsenal of methods of such isolation is insignificant and they are based mainly on knowledge of ecology of lichen species. So, it is known that in due course there is a change of properties of substratum, for example, bark of a linden (Tilia cordata) which in young age smooth and in mature is striated. Accordingly in due course varies also lichen species composition which living on this tree species.
It is necessary to consider also that urban environment substantially naturally is not, it is changed, and in many cases is created by the man, is especial in large megalopolises by which Moscow belongs also. Here frequently it is difficult to separate natural from anthropogenous. For example, the trees used for gardening. On the one hand it is the representatives of natural species of trees. With another - in many cases them cultivate in forest nurseries, and then transplanted in city.
For an interval of time between two terms of investigation (in beginning of 1990th and in 2006) young sapling mainly lindens and the maples planted at the end of 1980th in just built up inhabited files on periphery of city (Bibirevo, Mar'ino etc.) have turned to trees. Bark in the bottom parts of trunks on them from smooth became crack-striated. Accordingly, the probability of fastening on such substratum getting here of lichen propagules has increased. And if per 1988-1991 on a number of the sample plots 1 x 1 km in these areas lichens were absent or were submitted only by one of species though here there were no large sources of pollution in 2006 in these areas on the same trees lichens occur quite frequently. More often others recorded presence of Phaeophyscia orbicularis, Physcia adscendens, P. stellaris, Scoliciosporum chlorococcum, Xanthoria parietina thalli . Poverty lichen biota recently of built up urban territories they observed and in other cities, for example, in St. Petersburg (Malysheva, 2006).
In June, 2001 as a result of squally winds in northern areas of Moscow were lost some tens thousand trees, and their much greater number has damaged from windfall. Basically it there were 40-50- years poplars, birches, maples. Together with them could disappear and representatives of lichen species, there is once put up on their trunks as Cladonia caespitica, C. digitata, Parmeliopsis ambigua, P. hyperopta, Phaeophyscia ciliata, Physcia aipolia, Physconia grisea, which have not been occured in 2006 (tab. 3). On the other hand, as a result of this tornado in forests with density crown were formed extensive sites with rarefied the tree stand. That has resulted in change of a light condition and occurrence of thalli of heliophilous lichen species on the trees which kept in such sites. Or the species preferring more light habitats, began to meet here more frequently. It is the representatives of genera Caloplaca, Candelariella, Melanelia, Phaeophyscia, Physcia, Physconia, Xanthoria etc. The trunks of some trees as a result of tornado have changed a vertical standing on inclined. It has resulted in occurrence on them of thalli of species genera Cladonia and some other.
However most significant affect on current lichen biota composition in Moscow city was rendered with socio-economic factors. The results of investigation of lichens carried out at the end of 1980th reflect a state of lichen biota before catastrophic economic recession of a beginning 1990th. Many industrial enterprises which belonged to a category of stationary sources of air pollution of city, in middle 1990th or have stopped the activity, or considerably have lowered volumes of manufacture. Within 1990th a number of the enterprises were removed for a city boundaries, and on objects of power system the structure of used fuel has changed for the benefit of natural gas. All this has resulted in significant reduction of volume of emissions from stationary sources - with 177 000 tons in 1996 up to 93 100 tons in 2002 (Bityukova, Slobodskoi, 2002a, 2002b; Vasilenko et al., 2004). As a result of recession of activity of the industrial enterprises of city and adjacent regions, and also the improvements of quality of air in the countries of Western Europe, whence basically act to Moscow region air weights owing to transboundary transport, in Moscow acidity of rain water has decreased - if in 1987 the average annual value of pH of precipitation was 4.2, in 2002 - 6.25, highest for all previous period of measurement of precipitation acidity (1980-2002) on meteorological observatory of Moscow University (Yeremina, 2004). Moreover, in 2002 the acid rains (pH < 5.0) did not drop out. Up to 1991-1992 mineralization of precipitation increased and in 1991 the average annual value was 27.0 mg/l and concentration of sulphates in them was 10.9 mg/l. Per 2000-2001 the average value of precipitation mineralization has made 11.8 mg/l, and sulphates - 2.6 mg/l (Yeremina, 2004).
For these years has increased considerably number of automobiles in city and now it exceeds 3 million. Accordingly ratio between amount of emissions from stationary and mobile sources of pollution has changed considerably – quota first was steadily reduced from 41 % in 1986 up to 6-8 % in 2001-2005 (Moskva v zifrakh, 1987; Gosudarstvennyi doklad …, 2006). The structure of polluting substances in emissions has changed also. If in 1980 among pollutants prevailed COx (60 % from total emissions), NOx (14 %), hydrocarbons (13 %), SO2 (9 %), now the atmospheric air of city is most polluted by NOx, benzapilene, ammonia, formaldehyde. The tendency to appreciable growth of concentration of hydrocarbons, NO2 and NO, NH3, and chloride hydrogen is observed. The increase of concentration on first three parameters is connected with emissions from a vehicle. Thus, among air pollutants of city has decreased the amount of sulphur dioxide but considerably has increased amount of compounds of nitrogen in the form of ammonia and oxides.
However to consider nitrogen and its compounds as pollutants of environment it is very difficult, because this element is the main component of an atmosphere (more than ѕ its volume). It is a part of all organic substances and participates in all vital processes. Molecular nitrogen of an atmosphere are capable to fix and to transform it into the form, suitable for use, only the cyanobacterial lichen species. Such species are not fixed on trees in Moscow yet. However it is known, that the optimum of fixing of nitrogen at these species occurs at value of pH > 5, i.e. by neutral or alkaline conditions (Shapiro, 1986, 1996).
Ammonium NH4+ one of sources of nitrogen is absorbed by lichens passively (Dahlman et al., 2004) but rates of uptake is large as this cation is adhesion to the negatively charged cell walls (Brown et al., 1994). However high concentration of ammonium can suppress development even of nitrophytes also. Thus the experiment with Xanthoria parietina which is common nitrophytic lichen species for Moscow city has shown that the long treatment of thalli this species by NH4Cl in concentration 0.69M resulted in irreversible negative changes both mycobiont and photobiont. At concentration 0.35M NH4Cl at first there was a suppression of activity of both components, but then observed recovery up to former levels of activity both fungi and green algae, and the restoration of algae activity was more faster than fungi. It has given the basis to the authors of research to make a conclusion that photobiont of this lichens is better adapted to high levels of nitrogen in the environment (Gaio-Oliveira et al., 2004).
Primary pollutants between compounds of nitrogen they consider first of all ammonia and nitric oxide. Ammonia will neutralize SO2 in the form SO42– partially to NH4HSO4 or completely to (NH4)2SO4, and nitric oxide is readily oxidized by ozone to NO2 (Seaward, 2004). Further of products of oxidation of nitrogen oxides (NOx) of an atmosphere are formed secondary pollutants and chemical compounds, in gaseous or aerosol state. Ammonia NH3 is emitted by a large number of sources such as volatilization from animal waste and synthetic fertilizers, biomass burning, losses from soils under native vegetation and agricultural crops, etc (Krupa, 2003). Its exchange with an atmosphere is similar to an exchange of CO2, in which the compensation point determines concentration, at which the clean exchange does not occur. This compensation point is exceeded in conditions of modern agriculture both animal husbandry and ammonia is deposites and accumulates (Krupa, 2003).Because they will be carried out the actions on reduction of emissions SO2 and NOx in an atmosphere, the ammonia NH3 becomes the important agent of environment eutrophication and actually its hypertrophication. Now last term describes processes of enrichment of lichen habitats by nutrients more adequately (Seaward, 2004).
The increase of trophication of lichen habitats in Moscow city descend from the local sources (emissions of a traffic, road and building dust etc.). About localness of eutrophication sources of habitats evidence the data to reduction of concentration of nitrogen compounds as ammonium NH4+ or nitrate NO3– in rain water. For example, in 1992 concentration of ammonium in precipitation was 1.69 mg/l and in 2001 – 0.45 mg/l (Yeremina, 2004).
As a result of decrease of threshold value of acid pollution of habitats and the increases of a share alkaline pollution in territory of city were reestablished the representatives of the rather large number of lichen species adapted to moderately acid substratum and also there was an expansion on all territory of nitrophytic species (Caloplaca cerina, C. holocarpa, Lecanora hagenii, Phaeophyscia orbicularis, Physcia adscendens, P. stellaris, Xanthoria parietina etc.). The expansion is recorded at species Scoliciosporum chlorococcum also which considered they as toxitolerante acidophyte (Ahti, Vitikainen, 1974; Wirth, 1991). This species earlier occured in city quite frequently also. This lichen in 2006 was recorded in all surveyed 336 squares 1 x 1 km of six transects whereas in 1988-1991 index frequency of this species on same transects was 41% (tab. 3). Probably, it is necessary to reconsider recognition of this species as acidophytic because in Great Britain it already refer to nitrophytes (Wolseley, James, 2002; Davies, 2007). The abundance of species Hypogymnia physodes (R2), Lecanora varia (R2), Lepraria incana (R2) etc. has increased also. They are common for rather acid substratum. Probably, for these species earlier there was a threshold level of acid pollution which was decreased that has allowed them to appear in a number of the urban sites earlier not colonized with them.
The changes revealed in Moscow correspond to the tendency of behaviour of lichens in the large cities of the Europe from the end 1970th – beginning 1980th (Kandler, Poelt, 1984; Hawksworth, McManus, 1989). It was the reaction of lichens on the Acts on air clean entered in some countries. The returning of some sensitive to sulfur dioxide species in composition of local lichen biotas was observed in connection with reduction of pollution of an atmosphere by this compound. Later have found out that a significant role in composition of formed biotas, and not only urban, began to play the nitrophytic lichen species (van Dobben, 1993; van Herk, 1999; Purvis et al., 2003; Wolseley et al., 2006; Davies, 2007; Isocrono et al., 2007). Expansion of nitrophytic species of epiphytic lichens connect they with reduction of levels of sulfur dioxide in surface air and with high values of pH of tree bark because of pollution by a dust of a various origin (roadside, building, coal), ashes and soot, ammonia, of nitrogen oxides (van Dobben, de Bakker, 1996; van Herk et al., 2003; Larsen et al., 2007). For example, in two towns of Giessen land (Germany) with 1985 for 2005 the concentration of sulfur dioxide in air has decreased almost on the order, and the value of pH of bark of a linden has increased from 2.9-3.1 in 1985 till 5.9-6.3 in 2005, at a poplar – from 4.6-3.8. till 5.6-5.7. The number of epiphytic lichen species in these towns for the same period has increased from 11-21 till 49-52 (Kirschbaum et al., 2006). In London (UK) in 1960 on all trees was fixed 32 lichen species whereas per 2002-2003 only on ash (Fraxinus excelsior) have recorded out of the representatives 74 species (Davies et al., 2007). A wide distribution and abundance nitrophytic lichens explain they also by global warming (van Herk et al., 2002).
It is possible to explain the expansion.of a number nitrophytic species (Phaeophyscia orbicularis, P. sciastra, Physcia adscendens etc.) on all surveyed in 2006 territory of Moscow also that at these species prevails vegetative reproduction. Most frequently occurring species Phaeophyscia orbicularis, the indicator of environment hypertrophication in Western Europe (Seaward, 2004), forms huge number of soredia sometimes covering all surface of thallus. These vegetative propagules are easily get detached from thallus and are distributed by a wind or rain water, streaming down on a trunk. The fall into of bark roughness or being adhered to a layer of a dust, they quickly transform in thalli. Quite often on one trunk of a tree it is possible to count tens and hundreds thalli of this species. Frequently they grow together, and then practically it is impossible to separate one thallus from another. In the beginning 1990th these species was recorded in the greatest number of squares 1 x 1 km (43 %) also that evidenced about high eutrophication of habitats. But those years within the limits of these squares its thalli occured much less frequently and the covering by them of trunks of trees was small. In 2006 abundance of species on the sampling units was high, and the covering of trunks of trees up to height 2.5 m sometimes amounted to 30 %. The presence at city has increased of Parmelia sulcata also, species which distinguished by wide ecological amplitude but also distributing by soredia.
However in Moscow has increased the index frequency the species at which soredia and isidia is not present, and they are distributed by spores. It is the representatives Caloplaca, Candelariella, Lecanora, Physcia stellaris, Xanthoria parietina etc. It is possible that it is connected that them mycobionts can form thalli with the representatives of different species of green algae. For example, the fungi Xanthoria parietina uses for formation of thallus the representatives of three species of green algae Trebouxia – T. arboricola, T. decolorans, T. irregularis (Golubkova, 1993). Also is established that this species is adapted as to high, and low concentration of nitrogen compounds. In Portugal the concentration of nitrogen in thalli of this species collected in 13 various habitats, varied from 11 up to 43 mg/g dw (Gaio-Oliveira et al., 2005) whereas it is common at lichens with green algae as photobiont this parameter makes < 20 mg/g (Palmqvist et al., 2002). Also representatives Caloplaca, Xanthoria contain pigment parietin by which presence explain bright yellow - orange colour them thalli. However this complex compound includes anthraquinone, active antioxidant which neutralise free radicals formed in thallus at influence of pollutants (Silberstein et al., 1996; Sochting, Lutzoni, 2003). Thus, in Moscow the presence at lichens propagules for vegetative reproduction does not give the special advantage to colonization new sites above species at which such adaptations are absent. They observed that in London also (Larsen et al., 2007).
However first, as a rule, within the limits of the sampling units occur more frequently and cover more significant surfaces of substratum. Probably it is connected to features of distribution and attaching of lichen diasporas. They depend as on amount and quality produced propagules and the suitability for attaching sites where these propagules fall. For epiphytic lichens Lobaria pulmonaria with vegetative propagules (soredia) in forests of Sweden was established that for its distribution on the large distances (up to 75 m) means amount of produced diasporas, and for attaching near to parent thallus is important the presence of suitable conditions on substratum where they have fall (Öckinger et al., 2005). If project these data on results received in Moscow it is possible to assume that everywhere and quite often abundance presence of Phaeophyscia orbicularis thalli on trees of city is connected that this species produces vast amount vegetative propagules spreading by a wind and water on many tens meters. Thalli of Xanthoria parietina, as a rule, are fertile, i.e. with apothecia from which spores from time to time are thrown out. But their number considerably concedes to amount of P. orbicularis soredias. Besides in habitats where these spores have fall there should be suitable species of green algae that was generated thallus. Therefore abundance X. parietina in Moscow is rather insignificant compare with Phaeophyscia orbicularis.
It was earlier noted the increase of frequency in city for the compared period of the representatives of the species Scoliciosporum chlorococcum highly tolerance against pollution considered as acidophyte – in 2006 it was recorded in all surveyed 336 squares 1 x 1 km to six transects whereas in 1988-1991 index frequency of this species on same transects was 41 % (tab. 3). In cities of Western Europe with decrease of levels of acid pollution and increase alkaline recorded they digression at similar on stability to pollution acidophytic species Lecanora conizaeoides (Bates et al., 2001; Purvis et al., 2003; Kirschbaum et al., 2006). In Moscow city Scoliciosporum chlorococcum considerably has expanded the presence that puts under doubt an belonging it to group of acidophytes. But, probably, the levels of acid pollution all the same yet are high, and a lot of time has passed not so, that nitrophytic species have replaced this lichens.
I have tried to establish when in Moscow began the colonization by lichens of sites where they were absent earlier. For this purpose on sites where at the end of 1980th – beginning 1990th lichens did not occur, the autumn of 2006 by me carried out measurements of diameters of thalli of frequently occurred lichens Phaeophyscia orbicularis, Physcia stellaris, Xanthoria parietina (tab. 10). The measurements were carried out in area of the Crimean quay (city centre), on the Andropov prospectus at Kashira highway, in the Kustanayi street (southeast of city), in area Mar'ino (southeast of city), on Rossoshanskaya street (south of city), in area Bibirevo (north of city). On these sites was selected till 100 trees with lichens on which I measured diameters of largest thalli on the given trunk.
| Site | Phaeophyscia orbicularis | Physcia stellaris | Xanthoria parietina |
| Crimean quay | 18 | 28 | 26 |
| Andropov pr. | 24 | 32 | 40 |
| Kustanayi street | 20 | 29 | 22 |
| Mar'ino | 18 | 22 | 20 |
| Bibirevo | 25 | 28 | 40 |
| Rossoshanskaya street | 27 | 45 | 42 |
These data give the basis to believe, that colonization by lichens of various areas of city took place at different times. In our case it is possible to note, that in peripheral parts of city in the "youngest" area Mar'ino lichens have appeared later, than in the areas which have been built up earlier. Approximately at the same time, as in Mar'ino, colonization by lichens of city centre has begun also. The colonization of sites where lichens were absent before, possibly, proceeded cyclically. At first here were fixed single individuals of separate species nowadays submitted by largest thalli on the sizes. A bit later these individuals was capable to generate diaspores which were distributed in immediate proximity from a place of attaching of the primary arrivals. It needed some years on end which there was attaching the representatives of species on the next trees, fast increase of number of individuals, to what being evidence the increased amounts of thalli with smaller values of diameters. A bit later, probably, there will come some stabilization as species composition and number of individuals.
The diameter of thalli of the measured lichen species on average is increased by 2-3 mm per one year. It is possible to believe that active colonization by epiphytic lichens of the urban habitats where they were absent in the beginning 1990th began approximately in four – five years after 1992-1993 when began to be reduced of pressure of acid pollution of environment. This assumption is confirmed also with the data A.V. Pshchelkin (1998a, 1998b) which in second half 1990th recorded presence of some lichen species on sites where they were absent at the end of 1980th.
However at predicting possible future changes it is necessary to be considered that even if absolutely to stop receipt in an atmosphere anthropogenous SO2 and NOx, and the level of ammonia NH3 will not increase, that is doubtful as its source is the animal industries and in this case value of pH of precipitation will be about 5.0 (Yeremina, 2004). Accordingly trophication of epiphytes habitats will increase due to a mineral and organic dust deposited on trunks and branches of trees, and also of ammonia, which increases the contents of nitrogen in substratum and reduces acidity of last. The rain water, in itself already practically neutral, at stream on a trunk of a tree will be enriched by various elements which partially will get in lichen thalli. The species of neutral and eutrophic substratum will receive advantage before acidophytes and number of species last and also abundance can decrease. In Moscow while there is a process of implantation of species considered as acidophytes and their abundance is insignificant, and also take place expansion of group of nitrophytic lichens which are abundant and occur everywhere.
The previous period of lichens use as indicators of air pollution in cities and industrial areas passed in conditions of acid pollution of environment mainly by sulfur dioxide to which the lichens reacted rather sensitively. Nowadays quota of SO2 in pollution has decreased. However has increased amount of nitrogen compounds arrive in an environment. There is a question, whether can the lichens in new conditions to represent itself as reliable indicators of air pollution? Or on them it will be possible to determine a degree of habitats trophication? Probably the recent and future studies will give the answers to these questions.
The author in the previous publications (Biazrov, 1996, 2002, 2005) called to not overestimate a role of lichens as bioindicators of environmental conditions. The empirically established indexes and the correlations with concentration of number of xenobiotics have local importance, and patterns established in England or the Alpes not always it is possible extrapolate on the European part of Russia. A little still it is known about influence one phytotoxicants at presence in environment others or about transformation of the toxicants under environmental effects. For example, the emissions of organic compounds can be formed by 16-20 products of transformation, among which there can be more toxic and dangerous, than initial substance (Malysheva, 1997). Namely the motors of modern motor-cars throw out many organic compounds as benzene, toluene, phenol which as a result of active chemical interactions can quickly transformed to substances more toxic than initial. Therefore results of experiments in controllable conditions of laboratories not always reflect processes occurring in concrete habitats of lichens. In general at planning, realization and generalization of materials on use of lichens as bioindicators are appropriate the moderate scepticism and real estimation of their qualities.
It is desirable cite this material as:
Biazrov L.G. 2007: Changes of Epiphytic Lichen Species Diversity of Moscow City – Comparison between the Surveys of 1988-1991 and 2006. http://www.sevin.ru/laboratories_eng/biazrov_msk_2006.html